News that new diabetes medtech for an implantable insulin pump has reached an early regulatory milestone in the US.

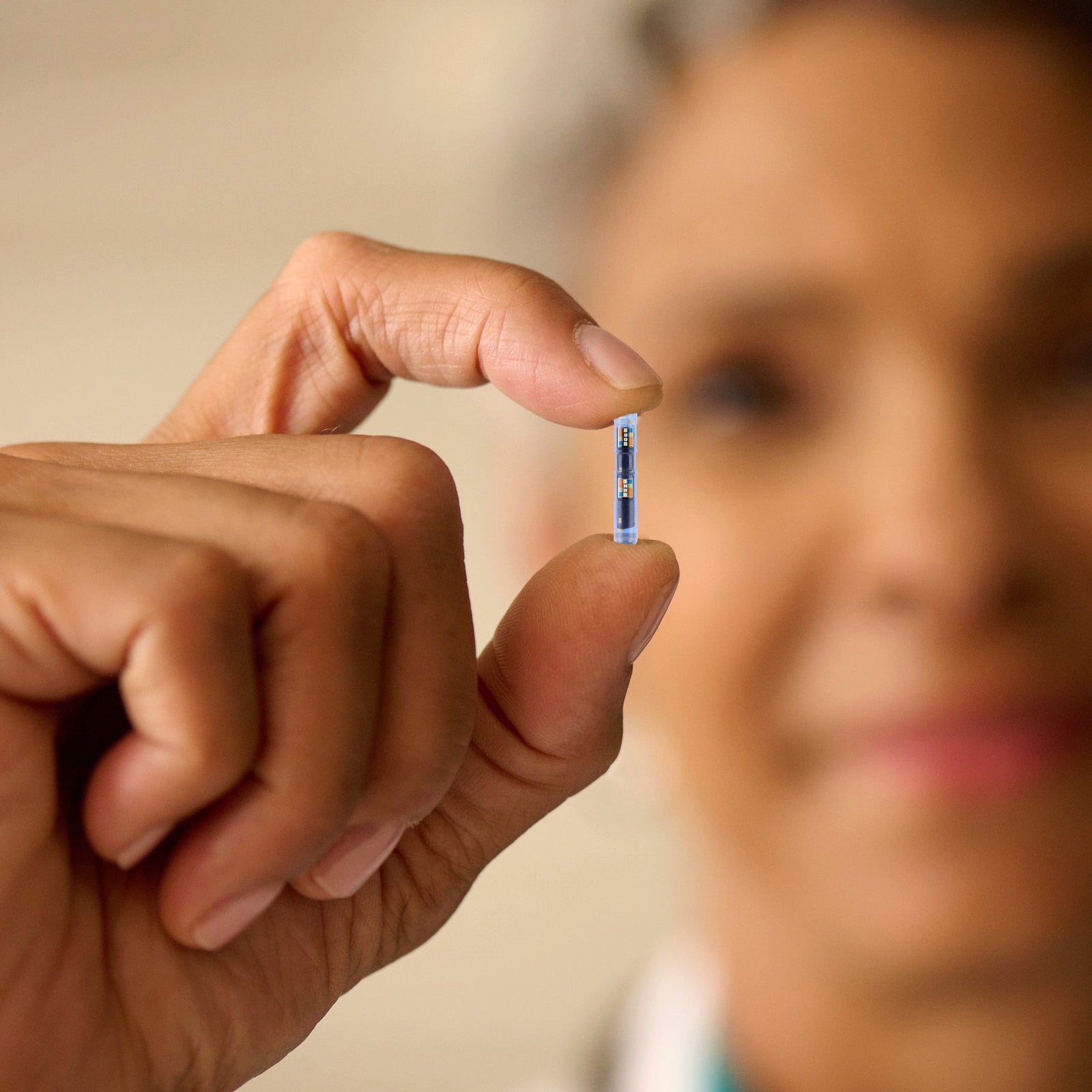
Image courtesy of Portal Diabetes
Portal Diabetes has announced that the U.S. Food and Drug Administration (FDA) has granted Breakthrough Device Designation to its implantable insulin pump system called the Portal Pump.
The company has also begun a Phase 1 clinical study of its investigational insulin formulation, Portal Insulin. The first injections in people have now taken place, marking an early step in development of the combined pump and insulin system.
What is the Portal Pump?
The Portal Pump is an implantable insulin pump designed for people with type 1 diabetes. Unlike external insulin pumps worn on the body, the device would be surgically implanted inside the abdomen.
The system under development includes:
• an implantable insulin pump
• continuous glucose monitoring (CGM) integration
• a concentrated and temperature-stable insulin formulation called Portal Insulin
The pump is designed to deliver insulin into the intraperitoneal space in the abdomen. This delivery route allows insulin to reach the liver directly, which is closer to how insulin works in people without diabetes.
Portal Diabetes says the aim is to combine implantable insulin delivery with modern CGM technology to create a fully automated insulin system that can manage glucose levels with minimal user input.
Phase 1 study of Portal Insulin begins
Portal Diabetes has begun a Phase 1 clinical study of Portal Insulin under an Investigational New Drug application with the FDA.
The first two participants recently received injections of the investigational insulin into the intraperitoneal space at a clinical site in San Diego, California.
This early study is designed to evaluate the safety of the insulin formulation and how it behaves in the body when delivered using the intraperitoneal route.
These injections represent the first clinical use of the company’s insulin formulation as part of the Portal Pump combination system.
What Breakthrough Device designation means
Breakthrough Device Designation from the FDA is intended for medical technologies that may provide significant advantages compared with existing treatments.
The designation provides several regulatory benefits during development, including:
• priority interaction with the FDA
• faster feedback during clinical trial planning
• eligibility for the Transitional Coverage for Emerging Technologies pathway, which can speed up Medicare coverage if the device is approved
The designation does not mean the device has been approved for use. Clinical trials and regulatory review are still required before the technology could become available to patients.
Previous implantable insulin pumps
Implantable insulin pumps are not a new concept in diabetes care.
In the 1980s and 1990s, Medtronic developed one of the first implantable insulin pump systems. These pumps were surgically implanted in the abdomen and delivered insulin directly into the intraperitoneal space.
Research showed that intraperitoneal insulin delivery could improve glucose control for some people because insulin reached the liver more directly.
Although the Medtronic implantable pump programme was later discontinued commercially, a small number of people in France still use the system through specialist hospital programmes. Medtronic continues to support these remaining patients.
When could the Portal Pump be available?
Portal Diabetes says clinical trials of the full Portal Pump combination system are expected to begin around the fourth quarter of 2027.
At present:
• the Portal Pump is not approved for use or sale in the United States
• Portal Insulin is an investigational drug and is not available commercially
Endocrinologists, surgeons and clinical centres interested in participating in future trials have been invited by the company to register their interest.
What this means for diabetes technology
Most modern insulin pump systems are worn externally and increasingly operate as hybrid-closed loop systems using CGM data.
The Portal Pump takes a different approach by placing the insulin pump inside the body and delivering insulin directly into the abdomen.
__________
Disclaimer
Blogs and publications on this website are independent of any involvement by medtech companies or diabetes related charities. To ensure there is no bias, we do not accept any products, freebies or other material from any medtech provider. Except where credited otherwise, all materials are copyright ©️Love My Libre Ltd.
What CGM is not associated or affiliated with Abbott or FreeStyle Libre. Content here and on our website what cgm.com does not constitute medical advice or replace the relationship between you and healthcare professionals nor the advice you receive from them.
The author of this blog has type 1 diabetes and uses a CGM on NHS prescription.


Leave a comment (all fields required). Please note, we are unable to respond to individual comments posted here.